Adding a heat will convert the solid into a liquid with no temperature change. 
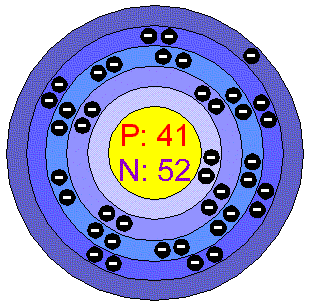
The melting point also defines a condition in which the solid and liquid can exist in equilibrium. The melting point of a substance is the temperature at which this phase change occurs. In general, melting is a phase change of a substance from the solid to the liquid phase. Since it is difficult to measure extreme temperatures precisely without bias, both have been cited in the literature as having the higher boiling point. Both the boiling points of rhenium and tungsten exceed 5000 K at standard pressure. In the periodic table of elements, the element with the lowest boiling point is helium. On the other hand, water boils at 350☌ (662☏) at 16.5 MPa (typical pressure of PWRs). A liquid at high pressure has a higher boiling point than when that liquid is at atmospheric pressure. A liquid in a partial vacuum has a lower boiling point than when that liquid is at atmospheric pressure. When considered as the temperature of the reverse change from vapor to liquid, it is referred to as the condensation point.Īs can be seen, the boiling point of a liquid varies depending upon the surrounding environmental pressure. The pressure at which vaporization (boiling) starts to occur for a given temperature is called the saturation pressure. Below the boiling point, the liquid is the more stable state of the two, whereas above the gaseous form is preferred. At the boiling point the two phases of a substance, liquid and vapor, have identical free energies and therefore are equally likely to exist. Any addition of thermal energy results in a phase transition. The liquid can be said to be saturated with thermal energy. The temperature at which vaporization (boiling) starts to occur for a given pressure is also known as the saturation temperature and at this conditions a mixture of vapor and liquid can exist together. The boiling point of a substance is the temperature at which this phase change (boiling or vaporization) occurs. In general, boiling is a phase change of a substance from the liquid to the gas phase. Note that, these points are associated with the standard atmospheric pressure. Irritation: Niobium dust causes eyes and skin irritation.Thermal Properties of Niobium Niobium – Melting Point and Boiling Point Medicine: Niobium is physiologically inert and hypoallergenic, meaning that it is an allergen to very few, for this reason it is used to make prosthetic arm.Superconducting Magnets: Niobium-germanium, niobium-tin, and niobium-titanium alloys are used as superconductor wires to make magnets which finds its use in MRI machines, NMR Spectroscopes, Large Hadron Collider (LHC) etc.Niobium is used in iron, cobalt, nickel based superalloys. Superalloys: This constitutes second largest use of the total niobium production.Niobium is sometimes used in very high quantity to produce highly wear and tear resistant machine elements. It is used for grain sizing, to retard recrystallization, and precipitation hardening. Steel industry: About 90% of the total niobium production is used in steel industries.Relevance in Chemical and Related Industries The same reaction without iron oxide is used to produce niobium metal directly but further processing has to be done to improve the grade. The products formed are aluminium oxide and ferroniobium, which is of industrial importance. Small amounts of sodium nitrate is added to enhance the reaction. Aluminothermic reaction: A mixture of aluminium oxide and niobium oxide is reacted with aluminium.This is reduced to obtain niobium in high purity. To obtain Niobium, first the ore is subjected to leaching using organic solvents then water is used to extract and then potassium fluoride is added to form a potassium fluoride complex. Extraction & Reduction: Mostly tantalum and niobium occur together in ores thus they are processed together and separated to obtain the element of desire.Niobium is capable of forming dielectric oxide layer.Niobium’s chemical properties are very similar to tantalum.On exposing niobium to air, an oxide layer is formed which is blue in colour.Niobium has only one stable isotope niobium-93.Niobium has quite high melting point but the density lower than refractory metals.Niobium has the greatest magnetic penetration depth of all elements.Niobium becomes a superconductor at cryogenic temperatures.The density of niobium is 8400 in S.I.Niobium is a light-grey, crystalline, and ductile transition metal.Niobium is said to be thirty-fourth most common element in the earth’s crust with concentration of 20 ppm. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
